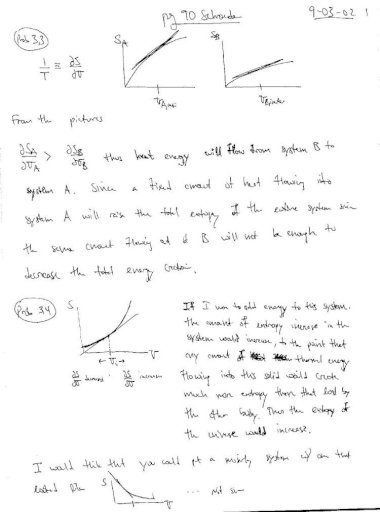
An introduction to thermal physics schroeder preview
In this book I have tried to do justice to both thermodynamics and statistical mechanics, without giving undue emphasis to either. To some extent the choice depends on what application areas one has in mind: Thermodynamics is often sufficient in engineering or earth science, while statistical mechanics is essential in solid state physics or astrophysics. Others put a strong emphasis on statistical mechanics, with its spectacularly detailed predictions and concrete foundation in atomic physics. Some prefer to cover only thermodynamics, it being less mathematically demanding and more readily applied to the everyday world. Physics instructors and textbook authors are in bitter disagreement over the proper content of a first course in thermal physics. This underlying explanation of thermodynamics, and the many applications that come along with it, comprise a subject called statistical mechanics. Then we can not only predict the properties of metals and other materials, but also explain why the principles of thermodynamics are what they are—why heat flows from hot to cold, for example. But to understand matter in more detail, we must also take into account both the quantum behavior of atoms and the laws of statistics that make the connection between one atom and 1023. These kinds of results, and the principles that generalize them, comprise a subject called thermodynamics. The maximum possible efficiency of an engine, working over a given temperature range, is the same whether the engine uses steam or air or anything else as its working substance. Liquids always boil more readily at lower pressure. Heat always flows spontaneously from a hot ob ject to a cold one, never the other way. Alternatively, we can measure the bulk properties of the metal (stiffness, conductivity, heat capacity, magnetization, and so on), and from these infer something about the particles it is made of.Some of the properties of bulk matter don’t really depend on the microscopic details of atomic physics. So instead, in thermal physics, we assume that the particles just jostle about randomly, and we use the laws of probability to predict how the chunk of metal as a whole ought to behave.
We can’t possibly follow every detail of the motions of all these particles, nor would we want to if we could. Anything big enough to see with our eyes (or even with a conventional microscope) has enough particles in it to qualify as a subject of thermal physics.Ĭonsider a chunk of metal, containing perhaps 1023 ions and 1023 conduction electrons. Examples include the air in a balloon, the water in a lake, the electrons in a chunk of metal, and the photons (electromagnetic wave packets) given off by the sun. Thermal physics deals with collections of large numbers of particles—typically 1023 or so.
